 History of previous stroke or transient ischemic attack is assigned 2 points. 6 Congestive heart failure, hypertension, diabetes mellitus, and age ≥75 years are each assigned 1 point. The CHADS 2 risk-scoring system is a well-validated method that utilizes five variables to determine the risk of cardioembolic stroke. 5įor early identification and better characterization of patients at high risk of developing strokes from AF, multiple risk scores have been developed. 4 In addition, AF-related strokes have poorer outcomes than non-AF-related strokes, including worse functional impairment, recurrence, and death. Patients with AF are susceptible to developing cardioembolic stroke, and are in fact five times as likely to suffer a stroke as patients without AF. Once available, edoxaban promises to be an important addition to the armamentarium of newer OACs for stroke prevention in patients with nonvalvular AF (NVAF). This has prompted the recent advent of newer agents including direct thrombin inhibitors like dabigatran, and factor Xa inhibitors like rivaroxaban and apixaban. 2, 3 The vitamin K antagonist (VKA) warfarin has traditionally been used for stroke prevention in these individuals, but effective use of this medication is encumbered by its delayed onset, narrow therapeutic window, numerous drug–drug and drug–food interactions, and need for regular monitoring. Patients with AF are at a high risk of developing cardioembolic strokes, leading to considerable morbidity and mortality. AF is the most common arrhythmia encountered in clinical practice. #Engage af timi 48 trialIt has been studied in a large Phase III trial (ENGAGE AF-TIMI 48), 1 and was recently reviewed and recommended for approval by a US Food and Drug Administration (FDA) advisory committee for its use for stroke prevention in patients with atrial fibrillation (AF).  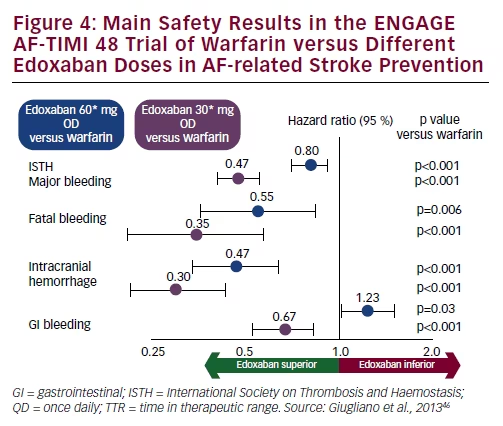
Keywords: atrial fibrillation, stroke prevention, novel oral anticoagulants, factor Xa inhibitors, edoxabanĮdoxaban belongs to the family of novel oral anticoagulants (NOACs), and is a direct factor Xa inhibitor. Once approved, it promises to provide another useful alternative to warfarin therapy. Edoxaban was recently reviewed by an FDA advisory committee and recommended as a stroke-prophylaxis agent. Treatment with a 30 mg or 60 mg daily dose of edoxaban was found to be noninferior to dose-adjusted warfarin in reducing the rate of stroke and systemic embolism in patients with nonvalvular atrial fibrillation, with a lower incidence of bleeding complications and cardiovascular deaths. 
Edoxaban is the latest oral direct factor Xa inhibitor studied in the largest novel oral anticoagulant trial so far: ENGAGE AF-TIMI 48. Direct thrombin inhibitor dabigatran and factor Xa inhibitors like rivaroxaban and apixaban have already been approved by the US Food and Drug Administration (FDA) for stroke prevention in patients with nonvalvular atrial fibrillation. Novel oral anticoagulant agents have recently become available as viable alternatives for warfarin therapy. Although effective, warfarin use is fraught with multiple concerns, such as a narrow therapeutic window, drug–drug and drug–food interactions, and excessive bleeding. Those at intermediate-to-high risk have traditionally been treated with therapeutic anticoagulation with warfarin for stroke prevention. As a result, atrial fibrillation patients are risk-stratified using the CHADS 2 or CHA 2DS 2-VASc scoring systems. It is responsible for significant morbidity and mortality from cardioembolic complications like stroke. Division of Cardiology, Department of Internal Medicine, University of California, San Francisco, Fresno, CA, USAĪbstract: Atrial fibrillation is the most common arrhythmia in the elderly. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
